The application of extreme pressure dramatically affects the chemical properties of xenon, so that it stops acting aloof and interacts with iron and nickel.
The paradox of the missing xenon might sound like the title of the latest airport thriller, but it’s actually a problem that’s stumped geophysicists for decades. New work from an international team including Carnegie’s Alexander Goncharov and Hanyu Liu, and Carnegie alumni Elissaios Stavrou and Sergey Lobanov, is chasing down the solution to this longstanding puzzle.
The mystery stems from meteorites, which retain a record of our Solar System’s earliest days. One type, called carbonaceous chondrites, contain some of the most-primitive known samples of Solar System material, including a lot more xenon than is found in our own planet’s atmosphere.
“Xenon is one of a family of seven elements called the noble gases, some of which, such as helium and neon, are household names,” said lead author Stavrou, now at Lawrence Livermore National Laboratory, about the team’s paper in Physical Review Letters. “Their name comes from a kind of chemical aloofness; they normally do not combine, or react, with other elements.”
Because xenon doesn’t play well with others, it’s deficiency in Earth’s atmosphere—even in comparison to other, lighter noble gases, like krypton and argon, which theoretical predictions tell us should be even more depleted than xenon—is difficult to explain.
That doesn’t mean many haven’t tried.
This research team—which also included Yansun Yao of the University of Saskatchewan, Joseph Zaug also of LLNL, and Eran Greenberg, and Vitali Prakapenka of the University of Chicago—focused their attention on the idea that the missing xenon might be found deep inside the Earth, specifically hidden in compounds with nickel and, especially, iron, which forms most of the planet’s core.
It’s been known for a while that although xenon doesn’t form compounds under ambient conditions, under the extreme temperatures and pressures of planetary interiors it isn’t quite so aloof.
“When xenon is squashed by extreme pressures, its chemical properties are altered, allowing it to form compounds with other elements,” Lobanov explained.
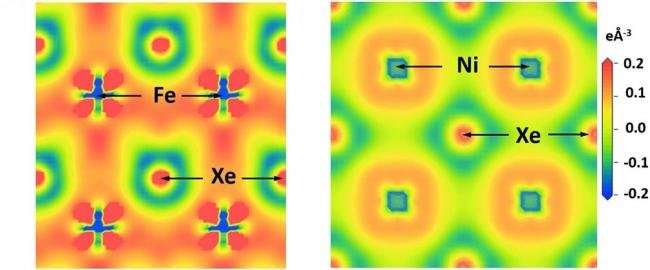
Using a laser-heated diamond anvil cell, the researchers mimicked the conditions found in the Earth’s core and employed advanced spectroscopic tools to observe how xenon interacted with both nickel and iron.
They found that xenon and nickel formed XeNi3 under nearly 1.5 million times normal atmospheric pressure (150 gigapascals) and at temperatures of above about 1,200 degrees Celsius (1,500 kelvin). Furthermore, at nearly 2 million times normal atmospheric pressure (200 gigapascals) and at temperatures above about degrees 1,700 degrees Celsius (2000 kelvin), they synthesized complex XeFe3 compounds.
“Our study provides the first experimental evidence of previously theorized compounds of iron and xenon existing under the conditions found in the Earth’s core,” Goncharov said. “However, it is unlikely that such compounds could have been made early in Earth’s history, while the core was still forming, and the pressures of the planet’s interior were not as great as they are now.”
The researchers are investigating whether a two-stage formation process could have trapped xenon in Earth’s early mantle and then later incorporated it into XeFe3 when the core separated and the pressure increased. But more work remains to be done.
Source: Phys.org






























Leave a Comment
You must be logged in to post a comment.